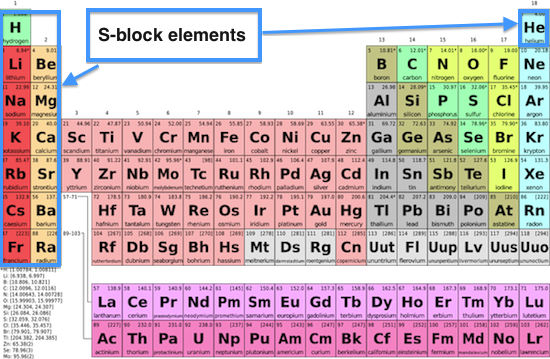
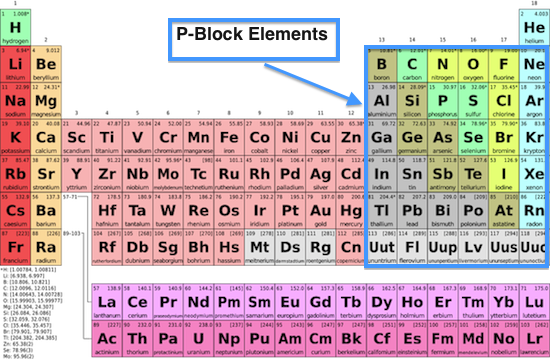
S Block Elements
Hydrogen
The Hydrogen element is commonly used for weather balloons due to its low density.
Alkali and Alkali Earth Metals
Alkali metals include Lithium (batteries), Sodium (salt), Potassium (fertilizer), Rubidium (GPS), Cesium (atomic clocks), and Francium (laser atom traps). These are the most active metals. Alkali Earth metals include Beryllium (alloy tools), Magnesium (cars), Calcium (milk), Strontium (flares), Barium (whitener for paper), and Radium (radiography). These Earth metals are the second most reactive elements.
P block elements
Boron Group
The Boron Group includes Boron (borax), Aluminum ( Earth's crust), Gallium (LED), Indium (anti corrosion agent), Thallium (rat poison). All of the elements have three electrons in their valence, Boron is the only one considered to be a metalloid and the rest are metals.
Carbon Group
The Carbon group includes Silicon (solar cells), Germanium (semiconductor for computer industry) , Lead (plumbing), Tin ( tin cans), and aluminum which replaced tin for can making. All of the elements have four electrons in its valence, and can form four bonds which is the most of any of the families.
Nitrogen Group
The Nitrogen family all have five electrons in its valence, the group is divided into nonmetals, semi metals, and metals. The elements include Nitrogen (explosives), Phosphorus (detergents), Arsenic and Antimony (alloys for batteries), and Bismuth (cosmetics).
Oxygen Group
The oxygen family can be found in nature in both free and combined states. The elements consists of Oxygen (breathe), Sulfur (matches), Selenium (X-ray), Tellurium ( cast iron), and Polonium (brush to get rid of dust in photographic film). These elements are willing to accept two electrons to form anions.
Halogens
The elements in group 17 create salts when react with metals. When the elements are in gaseous state, they are diatomic molecule which means they bond in two atoms. The elements include Fluorine (toothpaste), Chlorine (swimming pool water), Iodine (kill bacteria), Bromide and Iodide (photographic film).
Noble Gases
Noble gases are un reactive since they have a full outer shell of electrons. They are colorless and are Mon atomic, which means they exist as single atoms. The elements include Helium (balloons), Neon (glo-in-the dark signs), Argon (old style of light bulbs), Krypton (bright lamps), and Xenon (lighthouses).
D block Elements
Transition Metals
Transition metals are a collection of metals with similar properties. The elements of the first period are Scandium (furnace bricks), Titanium (white paint), Vanadium (drive shafts), Chromium (kitchenware), Manganese (plows), Iron (blood), Cobalt (Vitamin B-12), Nickel (heaters), Copper (wires), Zinc (fertilizer).
The next period of transition metals are Yttrium (furnace bricks), Zirconium (chemical pipelines), Niobium (MRI magnets), Molybdenum (armor plates), Technetium (medical diagnostic tracer), Rethenium (pen tips), Rhodium (relectors), Palladium (pollution control), Silver (jewelry), and Cadmium (fire sprinklers).
The last period of transition metals are Hafnium (plasma torch), Tantalum (mobile phones), Tungsten (abrasives), Rhenium (rocket engines), Osmium (needles), Iridium (spark plugs), Platinum (catalyst), Gold (coins), and Mercury (thermometers).
The next period of transition metals are Yttrium (furnace bricks), Zirconium (chemical pipelines), Niobium (MRI magnets), Molybdenum (armor plates), Technetium (medical diagnostic tracer), Rethenium (pen tips), Rhodium (relectors), Palladium (pollution control), Silver (jewelry), and Cadmium (fire sprinklers).
The last period of transition metals are Hafnium (plasma torch), Tantalum (mobile phones), Tungsten (abrasives), Rhenium (rocket engines), Osmium (needles), Iridium (spark plugs), Platinum (catalyst), Gold (coins), and Mercury (thermometers).
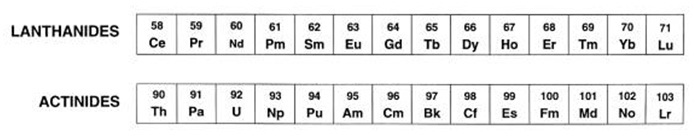
F block Elements
Inner Transition Metals
Rare Earths are chemical elements found in modern tech, computers, electronics, etc. They help innovate the modern tech by reducing its weight, reduce emission and many other contributions. The Rare Earths include the Lanthanides series, they include Cerium to reduce car emissions, Praseodymium for rare earth metals, Neodymium for infrared lasers, Promethium for watches, Samarium which can kill various cancers, Europium to prevent Euro money counterfeits, Gadolinium for MRI scans, Terbium fuel cells, Dysprosium for rarer earth magnets, Holmium for magnetic fields, Erbium for lasers, Thulium for military defenses, Ytterbium for cancer treatments, and Lutetium for determining the age of ancient items.
Lastly the Actinide series are radioactive metallic elements, certain elements are never found in nature since they are so short lived. These elements in the Actinide series include Thorium (gas lamp mantles), Protactinium (rad waste), Uranium (nuclear weapons), Neptunium (neutron detectors), Plutonium (nuclear reactor fuel), Americium (smoke detectors), Curium (mineral analyzers), Berkelium (rad waste), Californium (scientific instruments), and the rest are Einsteinium, Fermium, Mendelevium, Nobelium, and Lawrencium. Since they are short lived, no specific uses can be found.
Lastly the Actinide series are radioactive metallic elements, certain elements are never found in nature since they are so short lived. These elements in the Actinide series include Thorium (gas lamp mantles), Protactinium (rad waste), Uranium (nuclear weapons), Neptunium (neutron detectors), Plutonium (nuclear reactor fuel), Americium (smoke detectors), Curium (mineral analyzers), Berkelium (rad waste), Californium (scientific instruments), and the rest are Einsteinium, Fermium, Mendelevium, Nobelium, and Lawrencium. Since they are short lived, no specific uses can be found.